Surfactants are substances widely used in agricultural sprays to improve the solubility and mobility of pesticides across crops. This study investigates the micellar properties of two ionic surfactants sodium dodecyl sulfate (SDS) and tetramethylammonium tetrafluoroborate (TMATFB) with respect to their ability to solubilize the herbicide diuron in aqueous solution. Conductometric measurements were performed in aqueous media over a temperature range of 298 to 331 K to analyze the micellization behavior and evaluate the efficiency of solubilization. From the conductivity data the critical micelle concentration (CMC), and degree of ionization were obtained at various temperatures. Concentration and temperature effect on the CMC have been studied and the different thermodynamic parameters were evaluated. The critical micellar concentration (CMC) values of surfactants decreased with increasing temperature, indicating enhanced micelle formation under thermal influence. Additionally, the solubility of diuron varied significantly across different surfactant micelles concentration, suggesting that specific interactions occur between the surfactant head groups and the pesticide. The standard Gibbs free energy (∆G°) for the diuron–surfactant mixtures was attained to be negative throughout the study suggesting spontaneous micellization process. The enthalpy (∆H°) and entropy (∆S°) were also evaluated, offering additional insight into the thermodynamic driving forces involved. The obtained thermodynamic parameters showed that |TΔS°| is greater than |ΔH°|, suggesting that the micellization process is controlled by entropy.
| Published in | American Journal of Physical Chemistry (Volume 14, Issue 4) |
| DOI | 10.11648/j.ajpc.20251404.12 |
| Page(s) | 100-109 |
| Creative Commons |
This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited. |
| Copyright |
Copyright © The Author(s), 2025. Published by Science Publishing Group |
Diuron, Herbicide, Surfactants, Solubilisation, Water
Product | Formula | Molar mass (g mol−1) | WS (25 °C) (mg L−1) | MP (°C) | BP (°C) | Structure |
|---|---|---|---|---|---|---|
Diuron | C9H10Cl2N2O | 233.095 | 45 | 158 | 180 | |
SDS | CH3(CH2)11SO4Na | 288.38 | 1000 | 206 | NF | |
TMATFB | (CH3)4NBF4 | 160.95 | NF | NF | NF |
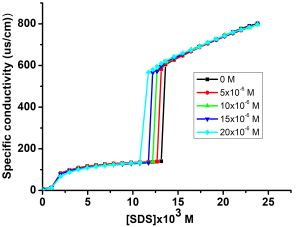
[Diur] (mM) | SDS | TMATFB | ||||
|---|---|---|---|---|---|---|
CMCa (mM) | αb (%) | ΔG° c (kJ mol-1) | CMCa (mM) | α b (%) | ΔG° c (kJ mol-1) | |
0 | 13.62 | 0.340 | -21.86 | 15.97 | 0.359 | -20.93 |
5 | 13.14 | 0.290 | -22.65 | 15.03 | 0.368 | -21.44 |
10 | 12.67 | 0.249 | -22.65 | 15.03 | 0.368 | -21.44 |
15 | 12.19 | 0.247 | -23.52 | 13.14 | 0.471 | -20.47 |
20 | 11.72 | 0.232 | -23.91 | 12.19 | 0.503 | -20.94 |
Surfactants | T (K) | cmcx103 M | XCMC | (%) | (KJ moL-1) | (KJ moL-1) | (KJ moL-1) |
|---|---|---|---|---|---|---|---|
SDS | 303 | 8.36 | 0.91 10-4 | 0.759 | -27.88 | 17.98 | 41.22 |
310 | 7.34 | 0.84 10-4 | 0.725 | -29.52 | 19.32 | 42.86 | |
317 | 6.42 | 0.80 10-4 | 0.533 | -35.31 | 23.25 | 50.50 | |
324 | 5.44 | 0.74 10-4 | 0.358 | -41.20 | 27.18 | 57.60 | |
331 | 4.46 | 0.71 10-4 | 0.323 | -43.50 | 28.98 | 59.78 | |
TMATFB | 303 | 12.69 | 2.29 10-4 | 0.260 | -23.149 | 5.25 | 38.49 |
310 | 12.19 | 2.41 10-4 | 0.318 | -23.512 | 5.53 | 39.31 | |
317 | 11.24 | 2.09 10-4 | 0.303 | -24.627 | 5.80 | 39.58 | |
324 | 10.30 | 2.07 10-4 | 0.336 | -25.082 | 6.27 | 41.22 | |
331 | 8.36 | 2.02 10-4 | 0.395 | -25.623 | 6.56 | 41.49 |
CMC | Critical Micellar Concentration |
| [1] | Williamson, S., Ball, A., Pretty, J. Trends in Pesticide Use and Drivers for Safer Pest Management in Four African Countries. Crop Prot. 2008, 27, 1327-1334. |
| [2] | Giller, K. E., Witter, E., Corbeels, M., Tittonell, P. Conservation Agriculture and Smallholder Farming in Africa: The Heretics’, View. Field Crops Res. 2009, 114, 23-24. |
| [3] | Bodian, E. T., Faye, C., Thiare, D. D., Diop, N. A., Diaw, P. A., Delattre, F., Coly, A., Giamarchi, P. Cyclodextrin-enhanced photo-induced fluorescence of tau-fluvalinate, molecular modelling of inclusion complexes and determination in natural waters. Anal. Methods. 2024, 16, 4347–4359. |
| [4] | Arias-Estevez, M., López-Periago, E., Martínez-Carballo, E., Simal-Gándara, J., Mejuto, J. C., García-Río, L. The mobility and degradation of pesticides in soils and the pollution of groundwater resources. Agric. Ecosyst. Environ. 2008, 123(4), 247-260. |
| [5] | Reichenberger, S, Bach, M., Skitschak, A., Frede, H. G. Mitigation strategies to reduce pesticide inputs into ground- and surface water and their effectiveness. a review Sci. Total. Environ. 2007, 384 (1-3), 1-35. |
| [6] | Bouwman, H. South Africa and the Stockholm Convention on Persistent Organic Pollutants. S. Afr, J. Sci. 2004, 100 (7), 323-328. |
| [7] | Jablonowski, N. D., Schäffer, A., Burauel, P. Still present after all these years: persistence plus potential toxicity raises questions about the use of atrazine. Environ. Sci. Pollut. Res. Int. 2011, 18, 328-359. |
| [8] | Diez, M. C. Biological aspects involved im the degradation of organic polluants. J. Soil Sci. Plant Nutr. 2010 10(3), 244- 267. |
| [9] | Bodian, E. T., Thiare, Diene Diegane, Bakhoum, J. Pierre., Mbaye, O. M. A., Diop, N. A., Diaw, P. A., Le Jeune, B., Coly, A., Giamarchi, P. Determination of Flumethrin and Tau-Fluvalinate Pyrethroid Insecticides in Surface and Groundwater by Photochemically Induced Fluorescence (PIF). Anal. Lett. 2022, 55, 1980–1996. |
| [10] | Aulagnier, F., Poissant, L., Brunet, D., Beauvais, C., Pilote, M., Deblois, C., Dassylva, N. Pesticides measured in air and precipitation in the Yamaska Basin (Quebec): occurrence and concentrations in 2004. Sci. Total. Environ. 2008, 394 (2-3), 338-48. |
| [11] | Coscollà, C., Colin, P., Yahyaoui, A., Petrique, O., Yusà, V., Mellouki, A., Pastor, A. Occurrence of currently used pesticides in ambient air of Centre Region (France), Atmos. Environ. 2010, 44 (32), 3915-392. |
| [12] |
Woodburn, A. T. Glyphosate: Production, pricing and use worldwide. Pest Manag. Sci. 2000, 56, 309-312.
https://doi.org/10.1002/(SICI)1526-4998(200004)56:4<309::AID-PS143>3.0.CO;2-C |
| [13] | Gianessi L. P. The Increasing Importance Herbicides in Worldwide Crop Production, Pest. Manag. Sci. 2013, 69, 1099-1105. |
| [14] | Burrows, H. D., Canle, L. M., Santaballa, J. A., Steenken, S. Reaction pathways and mechanisms of photodegradation of pesticides. J. Photochem. Photobiol. B. 2000, 7 (2), 71-108. |
| [15] | Thiare, D. D., Coly, A., Sarr, D., Khonte, A., Diop, A., Gaye Seye, M. D., Delattre, F., Tine, A., Aaron, J. J. Determination of the fenvalerate insecticide in natural waters by a photochemically-induced fluorescence method. Maced. J. Chem. Chem. Eng. 2015, 34 (2), 245-254. |
| [16] | Grant, R. J., Betts, B. W. Biodegradation of the synthetic pyrethroid cypermethrin in used sheep dip. J. Lett. Appl. Microbiol. 2003, 36 (3), 173-176. |
| [17] | Savadogo, P. W., Lompo, F., Coulibaly, K., Traore, O., Traore, A. S., Sedogo, M. P. A Microscom study of endosulfan degradation and its short-term effect on pH and biological parameters of cotton zones soils of Burkina Faso. J. Environ. Sci. Technol. 2009, 2, 12-21. |
| [18] | Ter Halle, A., Drncova, D., Richard, C. Hot transformation of the Herbicide Sulcotrione on Maize Cuticular Wax. Environ. Sci. Technol. 2006, 40 (9), 2989-2895. |
| [19] | Sambou, S., Thiare, D. D., Diaw, P. A., Sarr, I., Bodian, E. T., Mendy, A., Sarr, D., Gaye-Seye, M. D., Coly A. Analysis of diuron herbicide in Senegalese surface and groundwater depending on the soil depth by photochemically induced fluorescence (PIF). J. Iran. Chem. Soc. 2021, 18, 2389-2396. |
| [20] | Lu, Y. C., Guo, M. H., Mao, J. H., Xiong, X. H., Liu, Y. J., Li, Y. Preparation of core-shell magnetic molecularly imprinted polymer nanoparticle for the rapid and selective enrichment of trace diuron from complicated matrices. Ecotoxicol. Environ. Saf. 2019, 177, 66-76. |
| [21] | da Rocha, M. S., Nascimento, M. G., Cardoso, P. A., de Lima, P. L., Zelandi, E. A., de Camargo, J. L., de Oliveira, M. L. Cytotoxicity and regenerative proliferation as the mode of action for diuron-induced urothelial carcinogenesis in the rat. Toxicol. Sci. 2010, 113, 37-44. |
| [22] | da Rocha, M. S., Arnold, L. L., Pennington, K. L., Muirhead, D., Dodmane, P. R., Anwar, M. M., Battalora, M., de Camargo, J. L., Cohen, S. M. Diuron-induced rat bladder epithelial cytotoxicity. Toxicol. Sci. 2012, 130, 281-288. |
| [23] | Jeyraman, A., Karuppaiah, B., Chen, S. M., Huing, Y-C. Development of mixed spinel metal oxide (Co-Mn-O) integrated functionalized boron nitride: Nanomolar electrochemical detection of herbicide diuron. Physicochemical and Engineering Aspects is Colloids Surf. A: Physicochem. Eng. 2023, 666, 131278. |
| [24] | Saleck, M. L. O. C. O., Thiare, D. D., Sambou, S., Bodian, E. T., Sarr, I., Sarr, D., Diop, C., Gaye-Seye, M. D., Fall, M., Coly, A. Photochemically-Induced Fluorescence (PIF) and UV-vis Absorption Determination of Diuron and Metalaxyl in Well Water, Kinetic of Photodegradation and Rate of Leach Ability in Soils. Anal. Chem. Lett. 2019, 9, 806-815. |
| [25] | Ndiaye, N., Diouf, D., Faye, C. Sarr, A., Bakhoum, J-P., Thiare, D. D, Sarr, D., Coly, A., Giamarchi P. New automatic fluorescence monitoring system for insecticides in surface and groundwater. Applications to pyrethroids in the Niayes agricultural area of Senegal. Spectrochim. Acta A, 2026, 346, 126916. |
| [26] | Sarr, I., Thiare, D. D., Diaw, P. A., Bodàian, E. T., Sambou, S., K. kital, Mendy, A., Sarr, D., Fall, M., Delatre, F., Coly, A. Solubility and thermodynamic micellization studies of the insecticide chlorpyrifos in aqueous medium. J. Mater. Environ. Sci. 2022, 13, 70-81. |
| [27] | Kumar, D., Hidayathulla, S., Rub, M. A. Association behavior of a mixed system of the antidepressant drug imipramine hydrochloride and dioctyl sulfosuccinate sodium salt: Effect of temperature and salt. J. Mol. Liq. 2008, 271, 254-264. |
| [28] | Sristy, S. M. I. H., Mahbub, S., Alam, M. M., Rub, M. A., Hoque, M. A. Study of the interaction of tetradecyltrimethylammonium bromide with sodium dodecyl sulfate in aqueous/ urea solution at different temperatures: Experimental and theoretical investigatio. J. Mol. Liq. 2019, 284, 12-22. |
| [29] | Wang, Z., Zhao, F., Li, D. Determination of solubilization of phenol at coacervate phase of cloud point extraction. Colloids Surf. A: Physicochem. Eng. Asp. 2003, 216, 207-214. |
| [30] | Kumar, H., Chadha, C. Conductometric and spectroscopic studies of cetyltrimethylammonium bromide in aqueous solutions of imidazolium based ionic liquid 1-butyl-3-methylimidazolium tetrafluoroborate. J. Mol. Liq. 2015, 211, 1018-1025. |
| [31] | Gaillon, L., Lelièvre, J., Gaboriaud, R. Counterion Effects in Aqueous Solutions of Cationic Surfactants: Electromotive Force Measurements and Thermodynamic Model. J Colloid Interface Sci. 1999, 213, 287-297. |
| [32] | Galgano, P. D., El. Seoud, A. O. Micellar properties of surface-active ionic liquids: A comparison of 1-hexadecyl-3-methylimidazolium chloride with structurally related cationic surfactants. J. Colloid and Interf. Sci. 2010, 345, 1-11. |
| [33] | Thouvenet, M., Dousset, S. Compost effect on diuron retention and transport in structured vineyard soils. Pedosphere. 2015, 25 (1), 25-36. |
| [34] | Owoyomi, O., Ige, J., Soriyan, O. O. Thermodynamic of micellization of n-alkyl triphenyl phosphonium bromides: A conductimetric study. Chem. Sci. J. 2011, 25, 1-13. |
| [35] | Hu, X., Gong, H., Hollowell, P., Liao, M., Li, Z., Ruane, S., Liu, H., Pambou, E., Mahmoudi, N., Dalgliesh, R. M., Padia, F., Bell, G., Lu, J. R. what happens when pesticides are solubilised in binary ionic/zwitterionic-nonionic mixed micelles? J. Colloid. Interface. Sci. 2021, 586, 190-199. |
| [36] | Romanowski, M., Dynarowicz-Łątka, P. Semi fluorinated alkanes- Primitive surfactants of fascinating properties, Advances. Colloid. Interface. Sci. 2008, 138 (2), 63-83. |
| [37] | Jezequel, D., Mayaffre, A., Letellier, P. Behavior of cationic surfactants in pure molten ethyl ammonium nitrate, and its mixtures with water, A 298K. Potentiometric study, J. Chim. Phys. 1991, 88, 391-404. |
| [38] | Sarr, I., Bodian, E. T., Sambou, S., Mendy, A., Thiare, D. D., Diaw, P. A., Gaye-Seye, M. D., Coly, A., Tine, A. Conductometric study of the interaction of insecticide Profenofos with cationic and anionic surfactants in aqueous medium, Int. J. Pure Appl. Chem. 2017, 15, 1-9. |
| [39] | Thiare, D. D., Khonte, A., Diop, A., Cisse, L., Coly, A., Tine, A., Delattre, F. Determination of ground excited state dipole moments of amino-benzimidazole by solvatochromic shiff methods and theoretical calculations, J. Mol. Liq. 2015, 211, 640-646. |
| [40] | Abram, T., Chfaira, R. Study of the ionic micellar solubilization of an organic pollutant, case of phenol, J. Mater. Environ. Sci. 2015, 6 (2) (2015), 491-498. |
| [41] | Ajmal-Koya, P., Din, K., Ismail, K. Micellisation and thermodynamic parameters of butane diyl-1,4 bis (tetradecyltrimethylammonium Bromide) gemini surfactant at different temperatures: effect of the addition of 2-methoxyethanol, J. Sol. Chem. 2012, 41, 1271-1281. |
| [42] | Batigoc, C., Akbas, H., Boz, M. Micellization behavior and thermodynamic parameters of 12-2-12 gemini surfactant in (water +organic solvent) mixtures, J. Chem. Thermodyn. 2011, 43 (9), 1349-1354. |
| [43] | Cisse, A. K., Diouf, N., Faye, C., Sarr, I., Sambou, S., Cisse, A., Sidibe, B., Ndiaye, A., Bodian, E. T., Thiare, D. D., Coly, A. Enhanced Solubilization of Insecticide Phosalone by Using Anionic and Cationic Surfactants in Aqueous Solution by Micellisation, Asian J. Chem. Sci. 2024, 14, 137-147. |
| [44] | Batigoc, C., Akbas, H., Boz, M. Micellization behaviour and thermodynamic parameters of 12-2-12 gemini surfactant in (water+organic solvent) mixtures, J. Chem. Thermodyn. 2011, 43 (9), 1349-1354. |
| [45] | Thakur, R. A., Dar, A. A., Rather, G. M. Investigation of the micellar growth of 1-dodecylpyridinium chloride in aqueous solution of phenol, J. Mol. Liq. 2007, 136 (1-2), 83-89. |
| [46] | Yaïch, B. J., Debbabi, K., Naejus, R., Damas, C., Coudert, R., Baklouti, A. Evaluation of the surfactant properties of a series of monoalkyl oligoethoxylated F-alkyl α-Bomoesters, J. Soc. Chim. Tun. 2009, 11, 109-117. |
| [47] | Brinatti, C., Mello, L, B., Loh, W. Thermodynamic Study of the Micellization of Zwitterionic Surfactants and Their Interaction with Polymers in Water by Isothermal Titration Calorimetry, Langmuir. 2014, 30, 21, 6002-6010. |
| [48] | Alexis, T. F. C., Pelagie, Y., Coffi, A. E., Valentin, W. D. Evaluation du comportement de quelques savons traditionnels en solution aqueuse: Determination de la concentration micellaire critique et de la temperature de Krafft, J. Applied. Biosci. 2014, 83, 7493-7498. |
| [49] | Thiare, D. D., Sarr, D., Delattre, F., Giamarchi, P., Atanasse Coly, A. Direct and reverse micellar-enhanced photo-induced fluorescence determination of fenvalerate in senegalese surface and groundwater, J. Photochem. Photobiol. A 2024, 456, 115841. |
APA Style
Sambou, S., Sarr, I., Diouf, N., Faye, C., Cisse, A. K., et al. (2025). Enhanced Solubility of Herbicide Diuron in Aqueous Solution by Micellisation. American Journal of Physical Chemistry, 14(4), 100-109. https://doi.org/10.11648/j.ajpc.20251404.12
ACS Style
Sambou, S.; Sarr, I.; Diouf, N.; Faye, C.; Cisse, A. K., et al. Enhanced Solubility of Herbicide Diuron in Aqueous Solution by Micellisation. Am. J. Phys. Chem. 2025, 14(4), 100-109. doi: 10.11648/j.ajpc.20251404.12
AMA Style
Sambou S, Sarr I, Diouf N, Faye C, Cisse AK, et al. Enhanced Solubility of Herbicide Diuron in Aqueous Solution by Micellisation. Am J Phys Chem. 2025;14(4):100-109. doi: 10.11648/j.ajpc.20251404.12
@article{10.11648/j.ajpc.20251404.12,
author = {Souleymane Sambou and Ibrahima Sarr and Nehou Diouf and Coumba Faye and Abdou Khadre Cisse and Boubacar Sidibe and Aly Cisse and Keba Diongue and El Hadji Tombe Bodian and Diene Diegane Thiare and Atanasse Coly},
title = {Enhanced Solubility of Herbicide Diuron in Aqueous Solution by Micellisation},
journal = {American Journal of Physical Chemistry},
volume = {14},
number = {4},
pages = {100-109},
doi = {10.11648/j.ajpc.20251404.12},
url = {https://doi.org/10.11648/j.ajpc.20251404.12},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.ajpc.20251404.12},
abstract = {Surfactants are substances widely used in agricultural sprays to improve the solubility and mobility of pesticides across crops. This study investigates the micellar properties of two ionic surfactants sodium dodecyl sulfate (SDS) and tetramethylammonium tetrafluoroborate (TMATFB) with respect to their ability to solubilize the herbicide diuron in aqueous solution. Conductometric measurements were performed in aqueous media over a temperature range of 298 to 331 K to analyze the micellization behavior and evaluate the efficiency of solubilization. From the conductivity data the critical micelle concentration (CMC), and degree of ionization were obtained at various temperatures. Concentration and temperature effect on the CMC have been studied and the different thermodynamic parameters were evaluated. The critical micellar concentration (CMC) values of surfactants decreased with increasing temperature, indicating enhanced micelle formation under thermal influence. Additionally, the solubility of diuron varied significantly across different surfactant micelles concentration, suggesting that specific interactions occur between the surfactant head groups and the pesticide. The standard Gibbs free energy (∆G°) for the diuron–surfactant mixtures was attained to be negative throughout the study suggesting spontaneous micellization process. The enthalpy (∆H°) and entropy (∆S°) were also evaluated, offering additional insight into the thermodynamic driving forces involved. The obtained thermodynamic parameters showed that |TΔS°| is greater than |ΔH°|, suggesting that the micellization process is controlled by entropy.},
year = {2025}
}
TY - JOUR T1 - Enhanced Solubility of Herbicide Diuron in Aqueous Solution by Micellisation AU - Souleymane Sambou AU - Ibrahima Sarr AU - Nehou Diouf AU - Coumba Faye AU - Abdou Khadre Cisse AU - Boubacar Sidibe AU - Aly Cisse AU - Keba Diongue AU - El Hadji Tombe Bodian AU - Diene Diegane Thiare AU - Atanasse Coly Y1 - 2025/12/17 PY - 2025 N1 - https://doi.org/10.11648/j.ajpc.20251404.12 DO - 10.11648/j.ajpc.20251404.12 T2 - American Journal of Physical Chemistry JF - American Journal of Physical Chemistry JO - American Journal of Physical Chemistry SP - 100 EP - 109 PB - Science Publishing Group SN - 2327-2449 UR - https://doi.org/10.11648/j.ajpc.20251404.12 AB - Surfactants are substances widely used in agricultural sprays to improve the solubility and mobility of pesticides across crops. This study investigates the micellar properties of two ionic surfactants sodium dodecyl sulfate (SDS) and tetramethylammonium tetrafluoroborate (TMATFB) with respect to their ability to solubilize the herbicide diuron in aqueous solution. Conductometric measurements were performed in aqueous media over a temperature range of 298 to 331 K to analyze the micellization behavior and evaluate the efficiency of solubilization. From the conductivity data the critical micelle concentration (CMC), and degree of ionization were obtained at various temperatures. Concentration and temperature effect on the CMC have been studied and the different thermodynamic parameters were evaluated. The critical micellar concentration (CMC) values of surfactants decreased with increasing temperature, indicating enhanced micelle formation under thermal influence. Additionally, the solubility of diuron varied significantly across different surfactant micelles concentration, suggesting that specific interactions occur between the surfactant head groups and the pesticide. The standard Gibbs free energy (∆G°) for the diuron–surfactant mixtures was attained to be negative throughout the study suggesting spontaneous micellization process. The enthalpy (∆H°) and entropy (∆S°) were also evaluated, offering additional insight into the thermodynamic driving forces involved. The obtained thermodynamic parameters showed that |TΔS°| is greater than |ΔH°|, suggesting that the micellization process is controlled by entropy. VL - 14 IS - 4 ER -